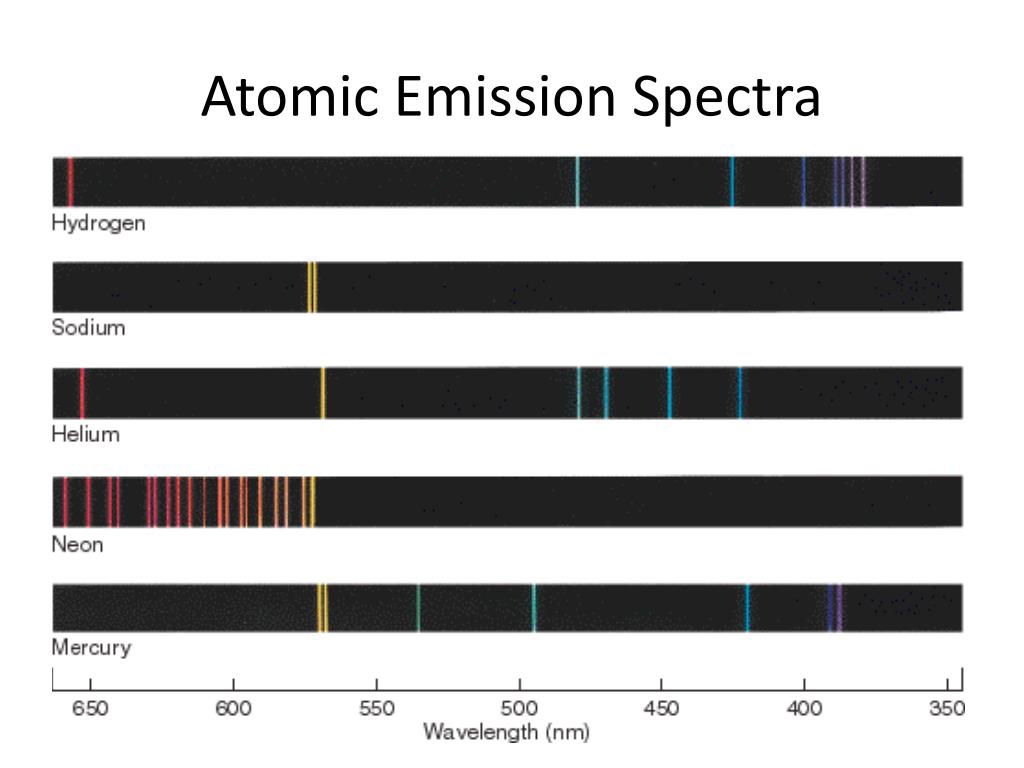 
In this model n = ∞ corresponds to the level where the energy holding the electron and the nucleus together is zero. If an electron jumps from a higher energy level to a lower energy level, energy will be. When an electron in an atom moves from high to low energy levels: Energy is emitted as electromagnetic radiation. An atomic emission spectrum is the pattern of lines formed when light passes through a prism to separate it into the different frequencies of light it contains. Gaussian fit to the data (black dotted line). 
The wavelengths corresponding to several transitions are shown. Figure 10.57 Valence shell energy level diagram for sodium. Common applications of atomic emission spectroscopy are in the analysis of trace elements in soils, water, metals, biological specimens, clinical specimens, food, physical. The sample to be tested could come from any number of sources. Each of these lines fits the same general equation, where n 1 and n 2 are integers and R H is 1.09678 x 10-2 nm-1. Figure 10.57 shows a portion of the energy level diagram for sodium, which consists of a series of discrete lines at wavelengths corresponding to the difference in energy between two atomic orbitals. Step 1: Atomic emission spectroscopy is used for the determination of the elemental composition of substances. Inset: magnified peak of the emission line at 546.0735 nm (literature value, entrance slit approx. Four more series of lines were discovered in the emission spectrum of hydrogen by searching the infrared spectrum at longer wave-lengths and the ultraviolet spectrum at shorter wavelengths. Data points (blue), literature peak positions of atomic emissions (red). If the light is passed through a prism or diffraction grating, it is split into its various colors. If a high voltage (5000 volts) is applied, the tube lights up with a bright pink glow. With Answer Key for Homeschool or Classroom (160 pgs) Spectrum on Amplify. It emits a specific wavelength distinct to that element. Atomic emission spectrum of a mercury vapour lamp as measured with the DIY CzernyTurner spectrometer. A hydrogen discharge tube is a slim tube containing hydrogen gas at low pressure with an electrode at each end. \hbar = 1.41 \hbar.\) is the Rydberg constant in terms of energy, Z is the atom is the atomic number, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus. Scarf Girl, they can Include claim, evidence, and reason in written.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
